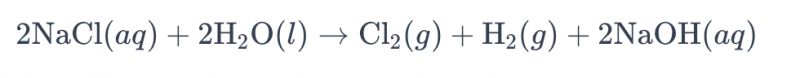Maitiro ekugadzira electrolyzing yemvura ine brine uchishandisa titanium electrodes kugadzira chlorine anowanzozivikanwa se "electrolysis yemvura ine brine." Mukuita uku, ma titanium electrodes anoshandiswa kubatsira kuita oxidation reaction yema chloride ions mumvura ine brine, zvichikonzera kugadzirwa kwegesi yechlorine. Muedzo wemakemikari wekuita kwemvura iyi ndewekuti:
Muequation iyi, maion echloride anooxidation paanode, zvichikonzera kugadzirwa kwegesi rechlorine, nepo mamorekuru emvura achidzikira pacathode, zvichiburitsa hydrogen gasi. Pamusoro pezvo, maion ehydroxide anodzikira paanode, zvichigadzira hydrogen gasi uye sodium hydroxide.
Sarudzo yemaelectrode etitanium inokonzerwa nekuti titanium inodzivirira ngura zvakanyanya uye inofambisa mvura, zvichiita kuti ikwanise kuita response yakagadzikana panguva yeelectrolysis isina ngura. Izvi zvinoita kuti maelectrode etitanium ave sarudzo yakakodzera yeelectrolysis yebrine.
Kugadziriswa kwemagetsi emvura ine saline kunowanzo da simba rekunze kuti ripe simba rekuita kwemagetsi. Simba iri rinowanzova simba remagetsi akananga (DC) nekuti mashandiro emagetsi anoda kuti magetsi aenderane negwara remagetsi, uye simba remagetsi reDC rinogona kupa gwara remagetsi rinoramba riripo.
Mukuita kwekushandisa electrolysing yemvura ine saline kuti igadzire gasi re chlorine, magetsi eDC ane voltage shoma anowanzo shandiswa. Voltage yemagetsi inoenderana nemamiriro chaiwo ekuita uye dhizaini yemidziyo, asi kazhinji inotangira pa2 kusvika 4 volts. Pamusoro pezvo, simba remagetsi riripo iparameter yakakosha inofanirwa kuongororwa zvichienderana nehukuru hwekamuri rekuita uye goho rekugadzirwa rinodiwa.
Muchidimbu, sarudzo yemagetsi ekugadzirisa mvura ine munyu zvinoenderana nezvinodiwa zvekuedza kana maitiro eindasitiri kuti zvive nechokwadi chekuti zvinoitwa zvinobudirira uye kuti zvigadzirwa zvinodiwa zviwanikwe.
Nguva yekutumira: Ndira-16-2024